Problem
Your company has 100’s or 1000’s of inspection, measuring and test instruments. It needs to track the status and whereabouts of these instruments and record any calibration activities. Someone needs to be notified of when they are due, coming due or overdue. We also need to track when the instrument was purchased, when it was calibrated, who calibrated it and what were the conditions and procedures used during calibration. This is a familiar problem for any company involved in any of the multiple of management systems out there. ISO 9001 quality management system standard requires a calibration system, TS16949, the automotive quality management system requires instrument tracking and calibration. There are lots of stand alone calibration programs available for a company to use, but why spend thousands when you can create a scalable system for recording, tracking, moving and calibrating your instruments within SharePoint
Solution
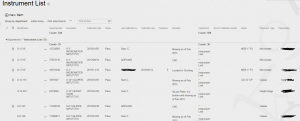
For starters create a list called “Instruments” add your required fields (Instrument ID, serial number, model, description, etc.). Once populated it will look like this;
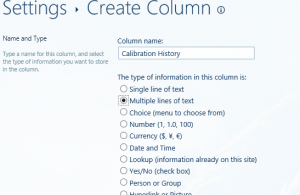
For a really basic tracker, from your list created above, add a field called calibration history;
Turn on versioning to the list, then turn on “append comments” to the “calibration history” field we just created.
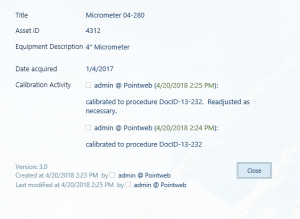
Every time a user creates an entry in “Calibration Activity” it leaves a time and date stamp of who made the entry.
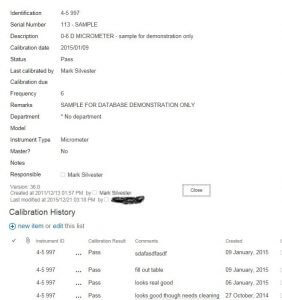
For those who want a more robust, scalable system with the instruments meta data and calibration history below, like this;
then read on. For starters, what we are going to do is create two lists; “Instruments information” and “Calibration Activities”. Once these are created we are going to link them together.
There are a few ways to do this linking;
- Create a lookup field in our calibration list that gets the instrument ID from the Instruments list.
- Create a workflow that copies the instrument ID to the calibration record
- Insert some simple code into our site that connects the “create new calibration record” action from within the instrument record that triggered it.
Each of these methods has is benefits and drawback.
Instrument record with calibration historyList one will hold the instruments static information such as such as purchased date, manufacturer, model, serial number, range, purchase price, etc. Whatever you want to have associated to the instrument. The thing about SharePoint is you can add new fields as you see fit.
Lets say the machinist would like an image of the instrument in the record, We can easily add that. Another possible requirement is to have a calibration procedure attached to the instrument information. We can easily add that with a link to the document library that holds our documented controlled instrument calibration procedures (I’ll save that for another blog post)
List two will hold our calibration history. Who calibrated it, when it was calibrated, what were the conditions at the time (temperature, humidity). Again, whatever you think is important to be recorded and associated to the instrument we are working on.


This is a great idea!